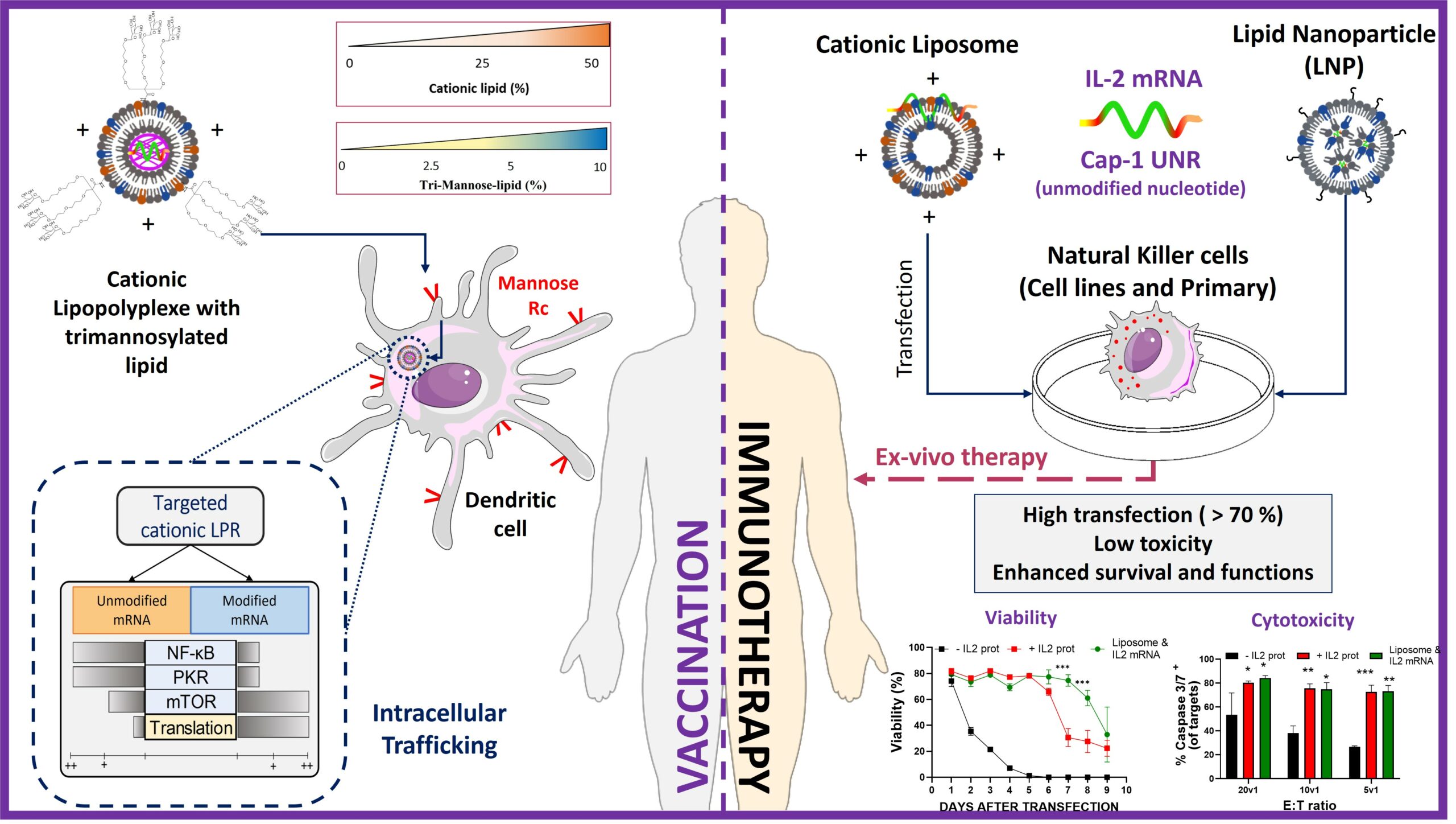
"Development of Versatile Lipid Formulations for Messenger RNA Delivery: Impact of Dendritic Cell Mannose Receptor Targeting and Natural Killer Cell Engineering".
A lire aussi
2024 May, 7 - El Hadji Cisse Defensis
23 April 2024
par Isabelle Frapart
July 1st-3rd "Skin Models in Cosmetic Science: Bridging Established Methods and Novel Technologies" - 3rd meeting
23 April 2024
par Isabelle Frapart
2024, May 24 - Seminar of Frank Schreiber
16 April 2024
par Isabelle Frapart