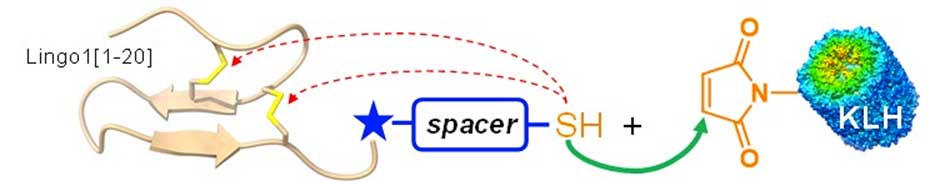
Her work “An efficient strategy to bioconjugate disulfide-containing peptides via thiol-maleimide chemistry while preserving their bridging pattern” was awarded at the “Chemical Biology Symposium 2024” conference organized by the Société Chimique de France.
A lire aussi
Le CBM recrute !
11 July 2025
par Isabelle Frapart
The CBM is recruiting!
11 July 2025
par Isabelle Frapart
Participation d'une équipe du CBM au Congrès national AMCSTI 2025
08 July 2025
par Isabelle Frapart